Risk Assessment on chemicals-For Better Understanding-3
Risk Assessment Methods
Risk assessment generally uses factors such as HQ (Hazard Quotient) and MOE (Margin Of Exposure). Both factors are based on the same concepts.
Risk Assessment HQ
HQ (Hazard Quotient) is calculated according to the following equation:
| HQ (Hazard Quotient) = | EHE (Estimated Human Exposure) |
| TDI (Tolerable Daily Intake) |
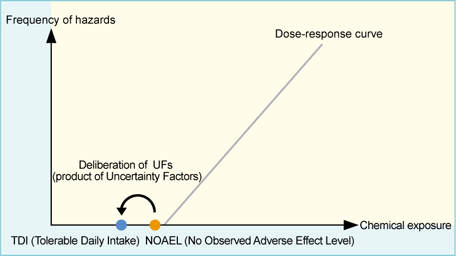
TDI (Tolerable Daily Intake)*1 is the amount of a chemical that can be taken daily by humans in a safe manner. It is calculated by dividing the NOAEL (No Observed Adverse Effect Level, determined in toxicity tests, etc.) by UFs (product of Uncertainty Factors)*2 and converted to the NOAEL for humans.
*1: See “NOAEL (No Observed Adverse Effect Level) and TDI (Tolerable Daily Intake)”
*2: See “UF (Uncertainty Factor)”
| TDI (Tolerable Daily Intake)= | NOAEL (No Observed Adverse Effect Level) |
| UFs (product of Uncertainty Factors) |
HQ (Hazard Quotient) compares the magnitude of EHE (Estimated Human Exposure)*3 with that of TDI (Tolerable Daily Intake). If HQ is larger than 1, i.e., if EHE exceeds TDI, there is risk. If HQ is smaller than 1, i.e., if EHE does not exceed TDI, there is no risk.
*3: See “EHE (Estimated Human Exposure)”
| If HQ (Hazard Quotient) ≥ 1 | Risk |
|---|---|
| If HQ (Hazard Quotient) < 1 | No Risk |
Risk Assessment using MOE (Margin Of Exposure)
MOE (Margin Of Exposure) is calculated according to the following equation:
| MOE (Margin Of Exposure) = | NOAEL (No Observed Adverse Effect Level) |
| EHE (Estimated Human Exposure) |
MOE (Margin Of Exposure) compares the magnitude of NOAEL (No Observed Adverse Effect Level) with that of EHE (Estimated Human Exposure). However the NOAEL is determined by toxicity tests, etc., so the calculation of MOE does not incorporate conversion into humans (it does not take uncertainty into consideration). So, MOE needs to be compared with UFs (product of Uncertainty Factors). If MOE is smaller than UFs, there is risk. If MOE is larger than UFs, there is no risk.
| If MOE (Margin Of Exposure) ≤ UFs (product of Uncertainty Factors) | Risk |
|---|---|
| If MOE (Margin Of Exposure) > UFs (product of Uncertainty Factors) | No Risk |
We may say that larger the UFs (product of Uncertainty Factors) are, the less reliable the assessment results become.
Differences in Risk Assessment methods
HQ (Hazard Quotient) includes UFs (product of Uncertainty Factors). So, whether or not the HQ is larger than 1 can be used as evaluation standards for Risk Assessments. MOE (Margin Of Exposure), on the other hand, does not include UFs (product of Uncertainty Factors). So, whether or not MOE is larger than UFs can be used as evaluation standards for Risk Assessments.
HQ (Hazard Quotient) is advantageous in that whether or not there is risk can be simply determined only by checking whether HQ is lager or smaller than 1.
Unlike HQ, MOE (Margin Of Exposure) does not include UFs (product of Uncertainty Factors).
MOE is advantageous in that it enables numerical analysis of uncertainty in Risk Assessment if used as a reference parameter, thus making differences by improving reliability, i.e., making it clear whether a judgment of “possible hazards” is attributable to a shortage of information (resulting in high UFs) or to the availability of information that is to some extent reliable.
column
*Within the framework of “General Chemicals Evaluation and Control Program” under NEDO Project, “Initial Risk Assessment Report” is prepared, and information related to risk assessment on chemicals is supplied.
Initial Risk Assessment Report
“Initial Risk Assessment Report” involves assessment of risks caused by chemicals in human health and ecotoxities and is prepared to determine the necessity of a more detailed risk assessment and the priority of chemicals requiring such assessments. Initial Risk Assessment used the MOE (Margin Of Exposure) as a factor.
*The explanation given above corresponds the influence on human health. When the influence on ecotoxicities is assessed, NOEC (No Observed Effect Concentration) instead of NOAEL (No Observed Adverse Effect Level), EEC (Estimated Environmental Concentration) rather than EHE (Estimated Human Exposure) is used.
Contact us
- Chemical Management Center, National Institute of Technology and Evaluation
-
Phone number:+81-3-3481-1977
Address:2-49-10 Nishihara, Shibuya-ku, Tokyo 1510066, Japan MAP
Contact Form